Package Insert for Extreme H2O Daily
PACKAGE INSERT
FOR THE
EXTREME H2O® DAILY
(hioxifilcon A)
Soft Contact Lens for Daily Disposable Wear
CAUTION: FEDERAL LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF
A LICENSED PRACTITIONER
Important: Please read carefully and keep this information for future use.
The following symbols may appear on the label or carton:

Lot Number

Manufacturer

CAUTION: U.S. Federal law restricts this device to sale by or on the order of a physician

Expiration Date (Use by date)

Sterile by steam
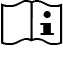
Consult instructions for use

Authorized Reprsentative in the European Community
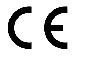
Indicates conformity with the essential health and safety requirements set out in European Directives
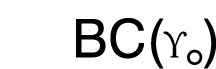
Base Curve

Diameter

Lens Power in Diopters
DESCRIPTION OF LENS
EXTREME H2O DAILY (hioxifilcon A) soft contact lenses are hemispherical shells and are available as spherical lenses of the following dimensions:
EXTREME H2O DAILY
PARAMETER
Diameter:
Center Thickness:
Base Curve:
Power Range:
VALUE
13.6 – 14.8
Varies with power
8.2 to 8.7
+6.00 to -10.00
The EXTREME H2O DAILY (hioxifilcon A) soft contact lens is fabricated from hioxifilcon A, which is a non-ionic, copolymer of 2-hydroxyethyl methacrylate (2-HEMA) and 2,3-Dihydroxypropyl Methacrylate (Glycerol Methacrylate, GMA) and cross-linked with ethylene glycol dimethacrylate (EGDMA). It consists of 41% hioxifilcon A and 59% water by weight when immersed in normal saline solution buffered with either sodium bicarbonate or sodium perforate. The lens is available with a blue visibility handling tint, phthalocyanato (2) – (copper).
The Physical/Optical properties of the lenses are:
The EXTREME H2O DAILY (hioxifilcon A) soft contact lens is fabricated from hioxifilcon A, which is a non-ionic, copolymer of 2-hydroxyethyl methacrylate (2-HEMA) and 2,3-Dihydroxypropyl Methacrylate (Glycerol Methacrylate, GMA) and cross-linked with ethylene glycoldimethacrylate (EGDMA). It consists of 41% hioxifilcon A and 59% water by weight when immersed in normal saline solution buffered with either sodium bicarbonate or sodium perforate. The lens is available with a blue visibility handling tint, phthalocyanato (2) – (copper).
The Physical/Optical properties of the lenses are:
PROPERTY / VALUE
Refractive Index: 1.515 (dry): 1.404 (hydrated)
Light Transmission – tinted: greater than 95%
Water Content: 59%
Specific Gravity: 1.308 (dry); 1.136 (hydrated)
Oxygen Permeability (Dk Value): 18 x 10-11 Fatt Units (cm2/sec)(ml O2/ml x mm Hg @ 35°C), revised Fatt method
ACTIONS
In its hydrated state, the EXTREME H2O DAILY (hioxifilcon A) soft contact lens, when placed on the cornea, acts as a refracting medium to focus light rays on the retina. The toric lens provides a more even surface over the highly uneven astigmatic cornea and thus helps to focus light rays on the retina.
CAUTION
Due to the small number of patients enrolled in clinical investigation of lenses, all refractive powers, design configurations, or lens parameters available in the lens material were not evaluated in significant numbers. Consequently, when selecting an appropriate lens design and parameters, the eye care practitioner should consider all characteristics of the lens that can affect lens performance and ocular health, including oxygen permeability, wettability, central and peripheral thickness, and optic zone diameter.
The potential impact of these factors on the patient’s ocular health must be carefully weighed against the patient’s need for refractive correction. Therefore, the continuing ocular health of the patient and lens performance on the eye should be carefully monitored by the prescribing eye care practitioner, since individual patient response may vary.
INDICATIONS
The EXTREME H2O DAILY (hioxifilcon A) spherical soft contact lens for daily wear is indicated for the correction of visual acuity in aphakic or not-aphakic persons with non-diseased eyes that are myopic or hyperopic. The lens may be worn by persons who exhibit astigmatism of 0.75 Diopters or less that does not interfere with visual acuity.
The EXTREME H2O DAILY (hioxifilcon A) toric soft contact lens for daily wear is indicated for the correction of visual acuity in aphakic or not-aphakic persons with non-diseased eyes that are myopic or hyperopic. The lens may be worn by persons who exhibit astigmatism of 10.00 Diopters or less.
Daily wear replacement schedules may vary from patient to patient and should be decided by the eye care practitioners in consultation with their patients. When prescribed for daily disposable use, no cleaning or disinfection is required. Lenses should be discarded upon removal.
CONTRAINDICATIONS
DO NOT USE the EXTREME H2O DAILY (hioxifilcon A) soft contact lenses when any of the following conditions are present:
• Acute or subacute inflammation or infection of the anterior chamber of the eye.
• Any eye disease, injury, or abnormality that affects the cornea, conjunctive, or eyelids.
• Severe insufficiency of lacrimal secretion (dry eyes).
• Corneal hypoesthesia (reduced corneal sensitivity), if not-aphakic.
• Any systemic disease that may affect the eye or be exaggerated by wearing contact lenses.
• Allergic reactions of ocular surfaces or adnexa that may be induced or exaggerated by wearing contact lenses or use of contact lens solutions.
• Allergy to any ingredient, such as mercury or thimerosal, in solution which must be used to care for EXTREME H2O DAILY soft contact lenses.
• Any active corneal infection (bacterial, fungi, or viral).
• If eyes become red or irritated.
• Patients unable to follow lens care regimen or unable to obtain assistance to do so.
WARNINGS
Patients should be advised of the following warnings pertaining to contact lens wear:
PROBLEMS WITH CONTACT LENSES AND CONTACT LENS CARE PRODUCTS COULD RESULT IN SERIOUS INJURY TO THE EYE. It is essential that you follow your eye care practitioner’s directions and all labeling instructions for proper use of your lenses and lens care products, including the lens case. EYE PROBLEMS, INCLUDING CORNEAL ULCERS, CAN DEVELOP RAPIDLY AND LEAD TO LOSS OF VISION; THEREFORE, IF YOU EXPERIENCE ANY DISCOMFORT, EXCESSIVE TEARING, VISION CHANGES, REDNESS OF THE EYE, IMMEDIATELY REMOVE YOUR LENSES AND PROMPTLY CONTACT YOUR EYE CARE PRACTITIONER.
Daily Wear lenses are not indicated for overnight wear, and patients should be instructed not to wear lenses while sleeping. Clinical studies have shown that the risk of
serious adverse reactions is increased when these lenses are worn overnight.
Studies have shown that contact lens wearers who are smokers have a higher incidence of adverse reactions than nonsmokers.
If a patient experiences eye discomfort, excessive tearing, vision changes, or redness of the eye, the patient should be instructed to immediately remove lens and promptly contact his or her eye care practitioner.
ALL CONTACT LENS WEARERS MUST RETURN FOR PERIODIC CHECK-UP VISITS AS RECOMMENDED BY THEIR EYE CARE PRACTITIONER.
PRECAUTIONS
• Clinical studies have demonstrated that contact lenses manufactured from hioxifilcon A are safe and effective for their intended use. However, the clinical studies may not have included all design configurations or lens parameters that are presently available in this lens material.
Consequently, when selecting an appropriate lens design and parameters, the eye care practitioner should consider all characteristics of the lens that can affect lens performance and ocular health, including oxygen permeability, wettability, central and peripheral thickness, and optic zone diameter.
The potential impact of these factors on the patient’s ocular health should be carefully weighed against the patient’s need for refractive correction; therefore the continuing ocular health of the patient and lens performance on the eye should be carefully monitored by the prescribing eye care practitioner.
• Fluorescein, a yellow dye, should not be used while the lens is on the eye. The lens absorbs this dye and become discolored. Whenever Fluorescein is used in eyes, the eyes should be flushed with a sterile saline solution that is recommended for in eye use. Wait at least one hour before replacing the lens. Too early replacement may allow the lens to absorb residual Fluorescein irreversibly.
• Before leaving the eye care practitioner’s office, the patient should be able to promptly remove the lens or should have someone else available who can remove the lens for him or her.
• Eye care practitioners should instruct the patient to remove the lens immediately if the eye becomes red or irritated.
Eye care practitioners should carefully instruct patients about the following care regimen and safety precautions:
• DO NOT use if the sterile blister package is opened or damaged. Always confirm the lens parameters printed on the multi-pack and on the individual lens package match the patient’s prescription. If there is a mismatch, the patient should not use the product.
• If the lens sticks (stops moving) on the eye, follow the recommended directions in Care for a Sticking (Non-Moving) Lens. The lens should move freely on the eye for the continued health of the eye. If non-movement of the lens continues, the patient should be instructed to IMMEDIATELY consult his or her eye carepractitioner.
• Always wash and rinse hands before handling lens. Do not get cosmetics, lotions, soaps, creams, deodorants, or sprays in the eyes or on the lens. It is best to put on lenses before putting on makeup. Water-based cosmetics are less likely to damage lenses than oil-based.
• Do not touch contact lenses with the fingers or hands if the hands are not free of foreign materials, as microscopic scratches of the lens may occur, causing distorted vision and/or injury to the eye.
• Carefully follow the handling, insertion, removal, and wearing instructions in the patient instructions for the EXTREME H2O DAILY (hioxifilcon A) soft contact lens and those prescribed by the eye care practitioner.
• Never wear lenses beyond the period recommended by the eye care practitioner.
• If aerosol products such as hair spray are used while wearing lenses, exercise caution and keep eyes closed until the spray has settled.
• Always handle lenses carefully and avoid dropping them.
• Avoid all harmful or irritating vapors and fumes while wearing lenses.
• Ask the eye care practitioner about wearing lenses during sporting activities.
• Inform your doctor (health care practitioner) about being a contact lens wearer.
• Never use tweezers or other tools to remove lenses from the lens container unless specifically indicated for that use. Pour the lens into the hand.
• Do not touch the lens with fingernails.
• Never rinse the lenses in tap water. Tap water contains many impurities that can contaminate or damage the lenses and may lead to eye infection or injury.
• Always discard lenses worn on a frequent/planned replacement schedule after the recommended wearing schedule prescribed by the eye care practitioner.
• When lenses are worn for daily disposable wear, the patient should be informed that no cleaning or disinfection is needed. Patients should always dispose of lenses when removed and have spare lenses or spectacles available.
• Always contact the eye care practitioner before using any medicine or medications in the eyes.
• Always inform the employer of being a contact lens wearer. Some jobs may require use of eye protection equipment or may require that the patient not wear contact lenses.
• As with any contact lens, follow-up visits are necessary to assure the continuing health of the patient’s eyes. The patient should be instructed as to a recommended follow-up schedule.
ADVERSE REACTIONS
The patient should be informed that the following problems may occur:
• Eyes stinging, burning, itching (irritation), or other eye pain.
• Comfort is less than when the lens was first placed in the eye.
• Feeling that something is in the eye such as a foreign body or scratched area.
• Excessive watering (tearing) of the eye.
• Unusual eye secretions.
• Redness of the eyes.
• Reduced sharpness of vision (poor visual acuity).
• Blurred vision, rainbows, or halos around objects.
• Sensitivity to light (photophobia).
• Dry eyes.
If the patient notices any of the above, he or she should be instructed to:
• IMMEDIATELY REMOVE LENS.
• If the discomfort or problem stops, then look closely at the lens. If the lens is in any way damaged, DO NOT PUT THE LENS BACK ON YOUR EYE. Place the lens in the storage case and contact your eye care practitioner. If the lens has dirt, an eyelash, or other foreign body on it, or the problem stops and the lens appears undamaged, the patient should thoroughly clean, rinse, and disinfect the lens then reinsert it.
• After reinsertion, if the problem continues, the patient should IMMEDIATELY REMOVE THE LENS AND CONSULT THE EYE CARE PRACTITIONER.
• When any of the above problems occur, a serious condition such as infection, corneal ulcer, neovascularization, or iritis may be present. The patient should be instructed to KEEP LENS OFF THE EYE AND SEEK IMMEDIATE PROFESSIONAL IDENTIFICATION of the problem and prompt treatment to avoid serious eye damage.
FITTING
Conventional methods of fitting contact lenses do apply to EXTREME H2O DAILY (hioxifilcon A) soft contact lenses. For a detailed description of the fitting techniques, refer to the Professional Fitting Guide, copies of which are available from:
Clerio Vision
7575 Commerce Court
Sarasota, FL 34243 USA
WEARING SCHEDULE
The eye care practitioner should determine the wearing and replacement schedule. Patients tend to over-wear the lenses initially. It is important not to exceed the initial wearing schedule. The eye care practitioner should emphasize the importance of adhering to the initial maximum wearing schedule. Regular check-ups, as determined by the eye care practitioner, are also extremely important.
Clerio Vision recommends that the lenses be discarded upon removal and replaced with new lenses daily.
EXTREME H2O DAILY (hioxifilcon A) soft contact lenses are indicated for daily wear. The maximum suggested wearing schedule for EXTREME H2O DAILY (hioxifilcon A) soft contact lenses is as follows.
SUGGESTED WEARING SCHEDULE
Days
1
2
3
4
5
6
7
Continuous Hours
3
3
4
4
6
6
8
Days
8
9
10
11
12
13
14 and after
Continuous Hours
8
8
10
12
14
15
All waking hrs
Studies have not been completed to show that EXTREME H2O DAILY (hioxifilcon A) soft contact lenses are safe to wear during sleep.
LENS CARE DIRECTIONS
Eye care practitioners should review with the patient that no cleaning or disinfection is needed with daily disposable lenses. Patients should always dispose of lenses when they are removed and have replacement lenses or spectacles available. Eye care practitioners may recommend a lubricating/rewetting solution, which can be used to wet (lubricate) the lenses while they are being worn to make them more comfortable. Note: Some solutions may have more than one function, which will be indicated on the label. Read the label on the solution bottle, and follow instructions.
• Care for a sticking (non-moving) lens:
If the lens sticks (cannot be removed), the patient should be instructed to apply 3 to 4 drops of the recommended lubricating or rewetting solution directly to the eye and wait until the lens begins to move freely on the eye before removing it. If non-movement of the lens continues after 15 minutes, the patient should IMMEDIATELY consult the eye care practitioner.
EMERGENCIES
The patient should be informed that if chemicals of any kind (household products, gardening solutions, laboratory chemicals, etc.) are splashed into the eyes, the patient should: FLUSH EYES IMMEDIATELY WITH TAP WATER FOR AT LEAST 15 TO 30 MINUTES AND IMMEDIATELY CONTACT THE EYE CARE PRACTITIONER OR VISIT A HOSPITAL EMERGENCY ROOM WITHOUT DELAY.
HOW SUPPLIED
Each EXTREME H2O DAILY (hioxifilcon A) soft contact lens is supplied sterile in a sealed blister pack containing buffered normal saline solution. The blister pack is labeled with the base curve, power, diameter, manufacturing lot number, and the expiry date of the lens. Do not use if the blister is damaged or the seal is broken.
REPORTING OF ADVERSE REACTIONS
All serious adverse experiences and adverse reactions observed in patients wearing EXTREME H2O DAILY (hioxifilcon A) soft contact lenses or experienced with the lens should be reported to:
Clerio Vision
7575 Commerce Court
Sarasota, FL 34243 USA
1-877-336-2482
To report serious adverse reactions for patients within the European Union, please contact the European Union Authorized representative:
Emergo Europe
Prinsessegracht 20
2514 AP The Hague
The Netherlands
CAUTION: FEDERAL LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF
A LICENSED PRACTITIONER
Manufactured and Marketed by
Clerio Vision
7575 Commerce Court
Sarasota, FL 34243 USA